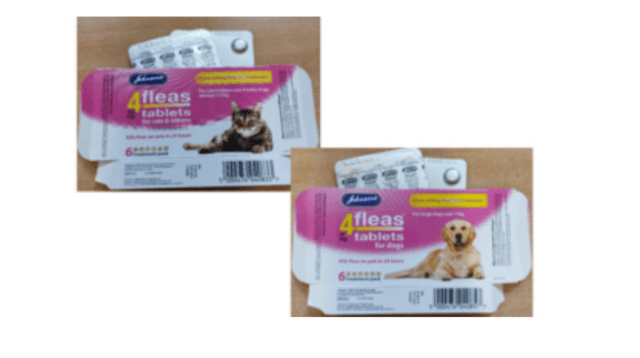
Wholesalers, retailers, and pet owners are advised to take note of a Class II recall initiated by Johnsons Veterinary Products. The affected product is Johnson’s 4Fleas 11.4mg Tablets for Cats & Kittens in the 6-tablet pack (D083) with Vm 00879/4060.
Reason for Recall: Some units from the batch intended for cats and kittens have been incorrectly packaged in cartons labelled as 4Fleas Tablets for Dogs (D085) with Vm 00879/4062. These cartons contain lower-strength 11.4 mg tablets meant for cats and kittens, posing a risk of ineffectiveness if administered to dogs. The recall specifically applies to batch number E154176 in 6-tablet packs.